Understanding Sacrificial Anodes on Ships
Corrosion is one of the greatest enemies of the ship and its machinery. It is also the toughest enemy to fight against, for the people working on the ship.
Iron is one substance which is used in abundance on the ship. From the main body of the ship to the smallest equipment used in operations, iron makes its presence felt in almost every type of equipment used onboard.
Iron is also the most corrosion-prone material when it comes in contact with air and water. A ship is continuously in contact with water and moisture-laden winds which makes it highly susceptible to corrosion. The outer body of the ship (mainly hull) is continuously in contact with water, making it extremely vulnerable to corrosion.
It is for this reason sacrificial anodes are used to protect the parent material.
In this article, we will have a look at the working of sacrificial anodes on a ship.
It is to note that the sacrificial anodes which are protecting the parent material should lie higher in the electromotive series of galvanic series of metals.
Sacrificial Anodes:
Metal strips of top-order metals in the reactivity series serve as anodes and are installed for cathode protection. These are called as sacrificial anodes.
For example, zinc can preferably be employed to serve the purpose of a sacrificial anode for aluminium as well as iron in the process of electrolysis.
The anode is divided into two parts namely Anode Insert and the Anode body.
Out of these two, the one which serves as an anode in the process of electrolysis is termed an anode body, whereas the other is called as anode insert, which is used to securely fix the anode to the parent surface by using brackets, bolts or weld.
Parent surface is the surface which is to be protected against corrosion. In general, we can say that the part of the ship to be protected against corrosion is called parent surface or cathode, whereas the more reactive material covering on the ship’s part which acts as an anode is termed as a sacrificial anode.
Classification Of Anodes
Anodes are categorized on a variety of parameters. As per the classification of anodes on the basis of their shape there are six types:
- Flat or block-shaped
- Tear-drop anodes
- Cylindrical or semi-cylindrical
- Disc-shaped
- Bracelet anodes and Tubular anodes
According to the size of anodes, they can be of two types – small-sized anodes and large-sized anodes.
On the basis of a material – anodes are Zinc anodes and aluminium, anodes are preferred in the marine industry.
On the basis of anode mounting method – there are flush-mounted anodes or slender anodes or sometimes just stand-off anodes. The different shaped anodes are applicable to different situations.
The shape of the anode can be decided on the basis of factors such as the shape of the equipment or part to be protected; space availability and installation ease; accessibility of different shapes in different conditions; etc.
For example, cylindrical pipelines are protected using cylindrical or bell-shaped anodes.
Anode Securing
The anode can be secured or fitted to the surface to be protected by three common methods which are: by the bolt; welds or with the use of brackets or ties.
Among these three methods welding is the most efficient method which can maintain maximum electrical continuity and remain in closest contact with the parent surface.
But bolting and bracket connection is used at places which cannot be accessed by welding. There is also another benefit of using bolts and brackets for connection is that they are replaceable when failing to work properly or due to any other requirement.
How Sacrificial Anodes Work?
Sacrificial anodes work on the principle similar to electrolysis, according to which, if an anode and a metallic strip are dipped in an electrolytic solution, anode electron will dissolve and deposit over the metallic strip and make it a cathode.
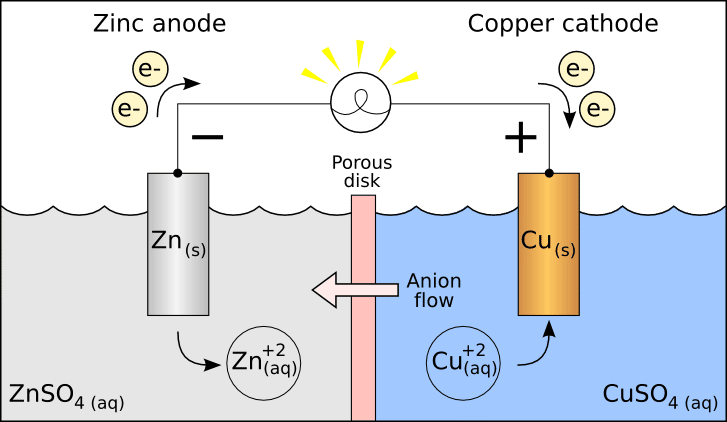
In the case of a ship, seawater acts as an electrolyte and transfers the electrons from the anode by oxidizing it over the steel plate and making a protecting layer.
If the metal is more active it will be easily oxidized and will protect the metallic compound by making it act as cathode. The anode will corrode first sacrificing itself for the other compound and it is thus called sacrificial anode.
Functions of Sacrificial anode:
Important functions of the sacrificial anode are as follows:
1) They are used to protect the main hull; ballast tanks and heat exchangers of a ship from corrosion.
2) Close the bottom of the vessel, storage chests are provided to cater to storage requirements by the sailor. In these sea chests too, sacrificial anode protection is provided to save them from corrosion.
Electromotive series or galvanic series metals
Anode materials
Magnesium(Mg)
Aluminium(Al)
Zinc (Zn)
Chromium(Cr)
Iron(Fe)
Nickel(Ni)
It can be seen from the table that for protecting iron any material above in the series are useful. These metals are preferred because they are easy and cheap to replace the anodes rather than complete a large sheet of metal.
These anodes are used in various applications such as :
1) Protecting the ship’s hull.
2) Protecting the ballast tanks corrosion.
3) Protecting the heat exchangers.
4) Sea Chests
Most Commonly Used Anode Materials:
Materials arranged as per their decreasing reactivity are: Magnesium (Mg); Aluminium (Al); Zinc (Zn); Chromium (Cr); Iron (Fe) and Nickel (Ni).
The materials which are at the top of this reactivity series can be used as anodes for the materials which is at a comparatively lower level.
In most cases in the maritime industry, zinc and aluminium are used as a sacrificial anode to protect iron or steel hull of the ship.
Frequency Of Changing Anodes
Anodes have to be changed only after they are completely decayed. The frequency for changing of anodes depends on the application where the anodes have been used.
In case the anodes are attached to the ship’s hull, then they are to be checked during dry dock which takes place after 2 to 3 years. If the anodes are found completely corroded then anodes of bigger size should be fitted, for fully corroded means that the material used was of poor quality or a large amount of material is required to protect the hull.
Generally, sacrificial anodes are changed at every dry dock.
If sacrificial anodes are used for heat exchangers and it is found during an inspection that the anode left is only 10% then also it has to be changed.
How to Assess if the Anodes are Working Properly or Not?
Inspections are planned for checking the proper working of a sacrificial anode. The important things to be noted to specify that anode is working properly or not are as follow:
- If anode does not dissolve from the time of installation to some later time at inspection and the parent material keeps on corroding, then this anode is ineffective and should be replaced with some other more reactive or efficient anode material.
- If there is no proper electrical continuity then also anode does not work properly. Because of this, the parent metal starts getting corroded instead of the anodes. Then check for proper connection between electrolytic solution and metal strips during the installation of setup.
Advantages of Sacrificial Anodes:
The main advantages of fitting sacrificial anodes to protect the ship and ship assembly are briefed as follows:
- These sacrificial anodes work without any external power source
- Installation is not at all any problem for sacrificial anodes. They are easy to install with the help of bolts, brackets or welds
- Inspection of these anode materials is quite easier
- They upturn the durability or service life of the ship by reducing corrosion attacks on it.
Disadvantages of Sacrificial anodes:
There are also certain disadvantages of sacrificial anodes along with the above advantages. The major disadvantages of Sacrificial anodes are briefed as under:
- Sometimes the material to be used as an anode is unable to work effectively due to lack of continuous electrical conductivity. This ineffective working of anode results in continuous corrosion of parent material.
- The current capacity increases with an increase in weight of anode, heavier the anode higher be the current capacity and high current capacity increases the effectiveness of electrolysis. The anode can work properly if it has an adequate current capacity. For ensuring this we need to fix heavier sheets of metal over the surface to be protected, which increases the overall weight of the ship and results in many problems.
Working and installation of Sacrificial anodes should be properly ensured so as to enhance the service life of the vessels to a certain extent. After properly inculcating the bans and boons of these anodes, these should be installed where required as per the pre-planned geometry and design of anodes.
If you liked this article, you may also like to read What is Marine growth preventive system (MGPS) ?.
Disclaimer: The authors’ views expressed in this article do not necessarily reflect the views of Marine Insight. Data and charts, if used, in the article have been sourced from available information and have not been authenticated by any statutory authority. The author and Marine Insight do not claim it to be accurate nor accept any responsibility for the same. The views constitute only the opinions and do not constitute any guidelines or recommendation on any course of action to be followed by the reader.
The article or images cannot be reproduced, copied, shared or used in any form without the permission of the author and Marine Insight.
Do you have info to share with us ? Suggest a correction

About Author
An ardent sailor and a techie, Anish Wankhede has voyaged on a number of ships as a marine engineer officer. He loves multitasking, networking, and troubleshooting. He is the one behind the unique creativity and aesthetics at Marine Insight.
Latest Marine Technology Articles You Would Like:
- 10 Harmful Effects Of Impure Air On Ship’s Machinery
- 10 Important Things to Check While Starting Fuel Oil Purifier on Ships
- 10 Noteworthy LNG-Powered Vessels
- 10 Points for Efficient Turbocharger Operation On Ships
- 10 Practical Tips to Handle Engine Room Pumps
- 10 Precautions to Take Before Operating Controllable Pitch Propeller (CPP) on Ships
Subscribe To Our Newsletters
By subscribing, you agree to our Privacy Policy and may receive occasional deal communications; you can unsubscribe anytime.














Hi Anish I am a marine student and at the moment in the final year. My project is on hull thickness testing near the sacrificial anode. Is is possible for you to post more information about this subject. It would be of great help for me.
Regards,
Saytan
Hi! I just want to ask a few questions. Actually these are not few question hehehe. Please help me
How oxidation take place?
what are the risks and consequence of oxidation?
how do you prevent it and protect specific cargo from oxidation?
Why does oxygen matters to you?
You are the OOW, you are due to load hazmat cargo. Explain
a.) what is the very first reference you must possess prior to loading to ensure you are receiving the cargo and stow them according to requirements?
b.) the PPE you must be wearing
c.during loading how do you conduct inspection to ensures cargo is apparently in good order?
Please use our forums- https://forums.marineinsight.com.
You will find most of the questions already answered there. If you did not find them, please post them as a question
That’s what I was looking for.I am glad that I came across such an article.I use to think how ships are there without getting rusted much .But after reading your article my doubt has been cleared.Thanks for sharing the best ones. Keep sharing such article.
@Jasmine: Glad you found the content useful. 🙂
Sacrificial Anode supplier
Hey Anish!
I really appreciate your efforts to write this awesome guide on Sacrificial Anodes on Ships. The way you expressed your thoughts and style of your writing is also nice,
I really liked your blog post, just going to share in my circle
@Steven: Thank you for your comment.
Can I ask some questions:
1. WHAT ARE THE APPROPRIATE MATERIAL USED AS ANODES FOR CONDENSER AND SEAWATER STRAINER CASING?
@Ryuk: Usually zinc or magnesium.
Hi Anish,
Thank you for a very informative article. On a related issue, are there any precautions that should be taken when using zinc anodes in an enclosed space (e.g. pipeline strainer) to prevent corrosion of the pipe and pumps?
This is very informative article.
@Laura: Thank you for your comment.
I got lot from this article. very informative.
Glad you found it useful 👍
Excellent! Reasonable in lenght and tick in information! Many thanks!!!
@Marcin: Glad you found the content useful 👍
Frequency of changing anode in db tanks
Frequency of changing anode in db tanks please advise